Ocean Acidification Lab
Bubbles Protocol
Title: The effects of composition of water on the absorption of Carbon Dioxide.
Topic Introduction: When carbon dioxide touches water, it creates ocean acidification. When that happens, that water then becomes acidic. With the ocean getting more & more acidic, it harms that animals, and the water. It affects the fishes, and many shellfish. The acidity has the ability to dissolve shells and weaken them, making shellfish an easy target to other predators.
Experimental Question: What will happen if we blow carbon dioxide into water?
Pre-Lab Questions: Bubbles Protocol (answer in complete sentences)
1. What gas are you blowing into the water?
I'm blowing carbon dioxide
2. What happens to the gas when you blow it into the water?
The gas absorbs the water
3. How are you measuring change in the water during this lab?
By the change of the color
4. What does measuring the pH of the water tell us?
It tells us how acidic it is
5. After studying the reactions above, how do you think carbonic acid will affect the pH of salt water? It will lower the pH of salt to become more acidic
Hypothesis: If I blow carbon dioxide into sea water, then it will become more acidic
Protocol:
Independent variable: The type of water
Dependent variable: The pH of the water after 30 seconds, 1 minute, 1 minute 30 seconds and 2 minutes.
Constants: The amounts of water and the type of wrap used in the experiment.
Preparation
1. Assign a role to each group member
2. Familiarize yourself with the Universal Indicator Color Chart (p. 7).
Control Trial
1. MATERIALS: Add 100 mL saltwater to a 500 ml beaker.
2. MATERIALS: Using your transfer pipet, add 4 drops of universal indicator. (Save your pipet for your next experiment!)
3. MATERIALS: Place a white sheet of paper under the beaker and record the initial color using the table on the data sheet.
4. MATERIALS: Stretch the piece of parafilm or saran wrap to completely cover the top of the beaker – then insert the straw by poking a hole through the parafilm.
5. RECORDER: Get the data sheet and prepare to record the color of the solution every 30 seconds, using the colors listed on the Universal Indicator Color Chart.
6. TIMER: Get timer ready to record for TWO MINUTES - Begin timing AT THE SAME TIME as breather begins blowing.
Ocean Acidification Bubbles Protocol
2BREATHER: Begin blowing through the straw into the water at exactly the same time as the timer begins timing. Breathe at a steady rate, exhaling only through straw. (Be careful to not inhale or suck on the straw!)
7. TIMER: Call out 30 second intervals.
RECORDER: As the timer calls out each 30 second interval, record the color at that time using the same color descriptions as the Universal Indicator Color Chart.
BREATHER: Exhale/blow at steady breathing rate for two full minutes.
8. RECORDER: Using the table on your data sheet and the Universal Indicator Color Chart, convert your color data to numbers, plot your data on the graph provided, and draw a line connecting the points to create a line graph.
9. GROUP: Decide on how you want to experiment by changing the variables. You will choose Protocol A, Protocol B, or Protocol C
Protocol B
1. MATERIALS: Add 100 mL saltwater to a 500 ml beaker.
2. MATERIALS: Place beaker on ice for about 3 minutes.
3. MATERIALS: Using your transfer pipet, add 4 drops of universal indicator. (Save your pipet for your next experiment!)
4. MATERIALS: Place a white sheet of paper under the beaker and record the initial color, using the table on the data sheet.
5. MATERIALS: Stretch the piece of parafilm to completely cover the top of the beaker – then insert the straw by poking a hole through the parafilm.
6. RECORDER: Get the data sheet and prepare to record the color of the solution every 30 seconds using the colors listed on the Universal Indicator Color Chart.
7. TIMER: Get timer ready to record for TWO MINUTES - Begin timing AT THE SAME TIME as breather begins blowing.
BREATHER: Begin blowing through the straw into the water at exactly the same time as the timer begins timing. Breathe at a steady rate, exhaling only through straw. (Be careful to not inhale or suck on the straw!)
8. TIMER: Call out 30 second intervals.
RECORDER: As the timer calls out each 30 second interval, record the color using the same color descriptions as the Universal Indicator Color Chart.
BREATHER: Exhale/blow at steady breathing rate for two full minutes.
9. RECORDER: Using the table on your data sheet and the Universal Indicator Color Chart, convert your color data to numbers, plot your data on the graph provided, and draw a line connecting the points to create a line graph.
Topic Introduction: When carbon dioxide touches water, it creates ocean acidification. When that happens, that water then becomes acidic. With the ocean getting more & more acidic, it harms that animals, and the water. It affects the fishes, and many shellfish. The acidity has the ability to dissolve shells and weaken them, making shellfish an easy target to other predators.
Experimental Question: What will happen if we blow carbon dioxide into water?
Pre-Lab Questions: Bubbles Protocol (answer in complete sentences)
1. What gas are you blowing into the water?
I'm blowing carbon dioxide
2. What happens to the gas when you blow it into the water?
The gas absorbs the water
3. How are you measuring change in the water during this lab?
By the change of the color
4. What does measuring the pH of the water tell us?
It tells us how acidic it is
5. After studying the reactions above, how do you think carbonic acid will affect the pH of salt water? It will lower the pH of salt to become more acidic
Hypothesis: If I blow carbon dioxide into sea water, then it will become more acidic
Protocol:
Independent variable: The type of water
Dependent variable: The pH of the water after 30 seconds, 1 minute, 1 minute 30 seconds and 2 minutes.
Constants: The amounts of water and the type of wrap used in the experiment.
Preparation
1. Assign a role to each group member
2. Familiarize yourself with the Universal Indicator Color Chart (p. 7).
Control Trial
1. MATERIALS: Add 100 mL saltwater to a 500 ml beaker.
2. MATERIALS: Using your transfer pipet, add 4 drops of universal indicator. (Save your pipet for your next experiment!)
3. MATERIALS: Place a white sheet of paper under the beaker and record the initial color using the table on the data sheet.
4. MATERIALS: Stretch the piece of parafilm or saran wrap to completely cover the top of the beaker – then insert the straw by poking a hole through the parafilm.
5. RECORDER: Get the data sheet and prepare to record the color of the solution every 30 seconds, using the colors listed on the Universal Indicator Color Chart.
6. TIMER: Get timer ready to record for TWO MINUTES - Begin timing AT THE SAME TIME as breather begins blowing.
Ocean Acidification Bubbles Protocol
2BREATHER: Begin blowing through the straw into the water at exactly the same time as the timer begins timing. Breathe at a steady rate, exhaling only through straw. (Be careful to not inhale or suck on the straw!)
7. TIMER: Call out 30 second intervals.
RECORDER: As the timer calls out each 30 second interval, record the color at that time using the same color descriptions as the Universal Indicator Color Chart.
BREATHER: Exhale/blow at steady breathing rate for two full minutes.
8. RECORDER: Using the table on your data sheet and the Universal Indicator Color Chart, convert your color data to numbers, plot your data on the graph provided, and draw a line connecting the points to create a line graph.
9. GROUP: Decide on how you want to experiment by changing the variables. You will choose Protocol A, Protocol B, or Protocol C
Protocol B
1. MATERIALS: Add 100 mL saltwater to a 500 ml beaker.
2. MATERIALS: Place beaker on ice for about 3 minutes.
3. MATERIALS: Using your transfer pipet, add 4 drops of universal indicator. (Save your pipet for your next experiment!)
4. MATERIALS: Place a white sheet of paper under the beaker and record the initial color, using the table on the data sheet.
5. MATERIALS: Stretch the piece of parafilm to completely cover the top of the beaker – then insert the straw by poking a hole through the parafilm.
6. RECORDER: Get the data sheet and prepare to record the color of the solution every 30 seconds using the colors listed on the Universal Indicator Color Chart.
7. TIMER: Get timer ready to record for TWO MINUTES - Begin timing AT THE SAME TIME as breather begins blowing.
BREATHER: Begin blowing through the straw into the water at exactly the same time as the timer begins timing. Breathe at a steady rate, exhaling only through straw. (Be careful to not inhale or suck on the straw!)
8. TIMER: Call out 30 second intervals.
RECORDER: As the timer calls out each 30 second interval, record the color using the same color descriptions as the Universal Indicator Color Chart.
BREATHER: Exhale/blow at steady breathing rate for two full minutes.
9. RECORDER: Using the table on your data sheet and the Universal Indicator Color Chart, convert your color data to numbers, plot your data on the graph provided, and draw a line connecting the points to create a line graph.
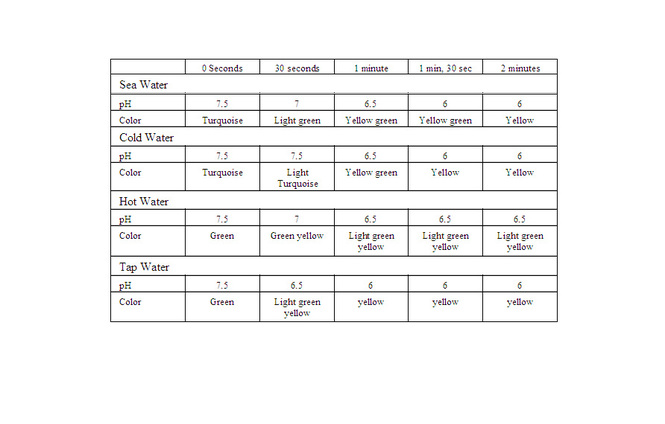
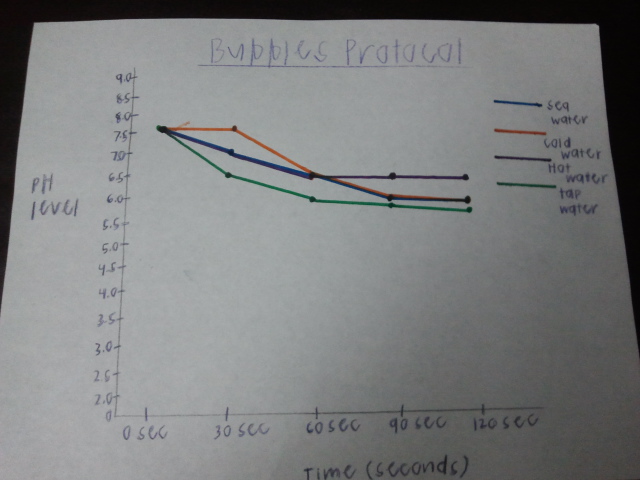
Data Table:
Graph:
Data Analysis: (Post-Lab Questions)
1. As you blew through the straw,what were you adding to the water and how did that
change the pH? As I blowing into the straw, I was adding carbon dioxide to the water which made the pH more acidic
2. What did the universal indicator tell us about the water?
The universal indicator told us the pH of the water
3. What does this tell us about the effects of carbonic acid in ocean water?
That when carbonic acid is in ocean water, the ocean becomes more acidic
4. Based on the results of your experimental protocol, which factor affects the pH of the
water most, temperature or salt? I think that temperature affects the pH because the acidity of each had different pH levels in the same amount of time
Conclusion: My initial hypothesis was correct. I stated that if I blow carbon dioxide into water, then it will become more acidic. With the universal indicator I able to see the pH level of the water and how it changed with more and more Carbon Dioxide over time. The more acidic the water got, the more the color started to change. From doing this experiment I see how carbon dioxide affects our ocean, and what will happen if this continues over the next couple years.
1. As you blew through the straw,what were you adding to the water and how did that
change the pH? As I blowing into the straw, I was adding carbon dioxide to the water which made the pH more acidic
2. What did the universal indicator tell us about the water?
The universal indicator told us the pH of the water
3. What does this tell us about the effects of carbonic acid in ocean water?
That when carbonic acid is in ocean water, the ocean becomes more acidic
4. Based on the results of your experimental protocol, which factor affects the pH of the
water most, temperature or salt? I think that temperature affects the pH because the acidity of each had different pH levels in the same amount of time
Conclusion: My initial hypothesis was correct. I stated that if I blow carbon dioxide into water, then it will become more acidic. With the universal indicator I able to see the pH level of the water and how it changed with more and more Carbon Dioxide over time. The more acidic the water got, the more the color started to change. From doing this experiment I see how carbon dioxide affects our ocean, and what will happen if this continues over the next couple years.
Shell Protocol
Central Question: How does a decrease in the pH of seawater affect the calcium carbonate shells of animals?
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
By combining calcium and carbon. Their shells are made out of calcium carbonate.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
I expect the shells to dissolve and break apart
3. What are sources of carbon dioxide and which of these sources are most likely to affect ocean pH? We humans exhaling, factories, and plants. I think us breathing affects it the most, because there are about 7.5 billion people in the world exhaling carbon dioxide every second and every minute of every day
Hypothesis: Shells Protocol- If I put shells into vinegar, then it will dissolve and break apart. Putting it into vinegar will dissolve it much faster the sea water
Protocol:
Independent variable: The type of liquid the shells are put in
Dependent variable: The mass of the shells
Constants: The beakers used, and the amount of liquid put in each
1. MATERIALS: Remove your two untreated shells from their bags.
2. MATERIALS: With a sharpie, label one shell “E’ for experimental and one shell “C” for control.
3. RECORDER: Using your data table record your group’s initial observations of the control and experimental shell characteristics.
4. MATERIALS: Find the mass of each of the shells.
5. RECORDER: Record the starting masses of both shells on your data table.
6. MEASURING: Pour 150 ml of vinegar into a 500 ml beaker and 150 ml of salt water into a second 500 ml beaker.
7. TIMER: Set the timer for 30 min.
8. MATERIALS: At the same time: Add the untreated, control shell “C” shell to salt water and the untreated, experimental shell “E” to the beaker of vinegar and start the timer.
9. RECORDER: In your data table, observe and record your group’s observations of what is happening to the shell while exposed to the vinegar over time. (at 0 minutes and 15 minutes) Ocean Acidification Shells Protocol
10. ***EVERYONE: Between observations of your shell in acid (vinegar), spend time observing and recording observations of shell characteristics for the pretreated (“Low Exposure” and “High Exposure”) shells.
11. MATERIALS: After 30 min use the tweezers to remove the shell from the vinegar and place on a paper towel. Dry the shell with a paper towel as best as you can.
12. MEASURING: Find the mass of both the control and the experimental shells.
13. RECORDER: Record the final masses and observations of the experimental and control shells after treatment.
14. EVERYONE: Decide how to test the strength of the shell. Options are:
a. Test the shell strength by dropping the shell from a height of 5 feet from the ground and then record the damage or breakage on your data table
b. Break the shell with text books to see how easily the shells break
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
By combining calcium and carbon. Their shells are made out of calcium carbonate.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
I expect the shells to dissolve and break apart
3. What are sources of carbon dioxide and which of these sources are most likely to affect ocean pH? We humans exhaling, factories, and plants. I think us breathing affects it the most, because there are about 7.5 billion people in the world exhaling carbon dioxide every second and every minute of every day
Hypothesis: Shells Protocol- If I put shells into vinegar, then it will dissolve and break apart. Putting it into vinegar will dissolve it much faster the sea water
Protocol:
Independent variable: The type of liquid the shells are put in
Dependent variable: The mass of the shells
Constants: The beakers used, and the amount of liquid put in each
1. MATERIALS: Remove your two untreated shells from their bags.
2. MATERIALS: With a sharpie, label one shell “E’ for experimental and one shell “C” for control.
3. RECORDER: Using your data table record your group’s initial observations of the control and experimental shell characteristics.
4. MATERIALS: Find the mass of each of the shells.
5. RECORDER: Record the starting masses of both shells on your data table.
6. MEASURING: Pour 150 ml of vinegar into a 500 ml beaker and 150 ml of salt water into a second 500 ml beaker.
7. TIMER: Set the timer for 30 min.
8. MATERIALS: At the same time: Add the untreated, control shell “C” shell to salt water and the untreated, experimental shell “E” to the beaker of vinegar and start the timer.
9. RECORDER: In your data table, observe and record your group’s observations of what is happening to the shell while exposed to the vinegar over time. (at 0 minutes and 15 minutes) Ocean Acidification Shells Protocol
10. ***EVERYONE: Between observations of your shell in acid (vinegar), spend time observing and recording observations of shell characteristics for the pretreated (“Low Exposure” and “High Exposure”) shells.
11. MATERIALS: After 30 min use the tweezers to remove the shell from the vinegar and place on a paper towel. Dry the shell with a paper towel as best as you can.
12. MEASURING: Find the mass of both the control and the experimental shells.
13. RECORDER: Record the final masses and observations of the experimental and control shells after treatment.
14. EVERYONE: Decide how to test the strength of the shell. Options are:
a. Test the shell strength by dropping the shell from a height of 5 feet from the ground and then record the damage or breakage on your data table
b. Break the shell with text books to see how easily the shells break
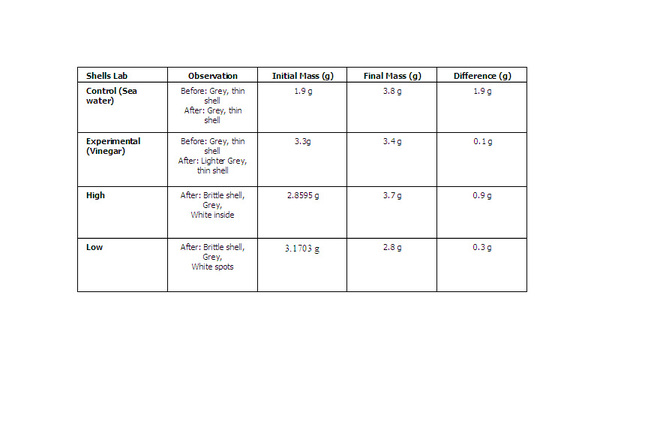
Data Table:
Analysis/Discussion of Data
1. When you immersed the shells in vinegar how did you know that a reaction was
happening? I knew there was a reaction happening because bubbling started to occur
2. How did observing the shells in vinegar relate to how animals are affected by a lower pH of ocean water? Testing these shells, showed an example of what happens to animals shells in the ocean
3. How would shelled organisms be affected by a lower pH of ocean water?
They would dissolve because of the low pH of ocean water
4. What are the primary functions of shell for these animals?
The primary functions of shells are to protect that from predators and for shelter
5. Does it cost the animal energy to rebuild or repair their shell?
Yes because they use more energy and nutrients causing them to use more energy
Conclusion: My initial hypothesis was true because putting shells into vinegar resulted into the mass of the shell to go down. The mass of the shell that was placed in regular sea water remained the same. From this experiment I learned that low pH levels in the ocean cause animal shells to dissolve. When these shells dissolve, these animals are more vulnerable to predators. With all these animals gone, the food chain won't be balanced and the rest might die out also.
1. When you immersed the shells in vinegar how did you know that a reaction was
happening? I knew there was a reaction happening because bubbling started to occur
2. How did observing the shells in vinegar relate to how animals are affected by a lower pH of ocean water? Testing these shells, showed an example of what happens to animals shells in the ocean
3. How would shelled organisms be affected by a lower pH of ocean water?
They would dissolve because of the low pH of ocean water
4. What are the primary functions of shell for these animals?
The primary functions of shells are to protect that from predators and for shelter
5. Does it cost the animal energy to rebuild or repair their shell?
Yes because they use more energy and nutrients causing them to use more energy
Conclusion: My initial hypothesis was true because putting shells into vinegar resulted into the mass of the shell to go down. The mass of the shell that was placed in regular sea water remained the same. From this experiment I learned that low pH levels in the ocean cause animal shells to dissolve. When these shells dissolve, these animals are more vulnerable to predators. With all these animals gone, the food chain won't be balanced and the rest might die out also.